What Is MET?
The MET gene is located in chromosome 7 in the cells of the human body. Within the MET gene are the instructions to produce the MET protein. The instructions for the MET protein is broken into segments called Exons. When all the Exons are properly executed in sequence, the normal MET protein is produced. As a note, the MET protein may also be referred to as c-MET, tyrosine-protein kinase MET or hepatocyte growth factor receptor (“HGFR”). In this discussion, we will refer to it as MET.
The MET protein is essential in normal embryonic development, organ development, and wound healing. The MET protein is also degraded within cells as part of its normal life cycle.
The MET protein penetrates the cell wall. There is a portion outside the cell that is the HGRF (hepatocyte growth factor receptor), an intercellular portion where the cell wall is penetrated and a portion internal to the cell that controls tyrosine kinase signaling for cell division. The tyrosine kinase portion of MET controls the enzymatic activity that acts as an on off switch for cellular growth and division.
In the case of cancer driven by MET, there is an alteration in the MET gene causing an error in the production of the MET protein that prevents its degradation or causes excessive tyrosine kinase activity. The result of this process is the cancer cells grow unchecked.
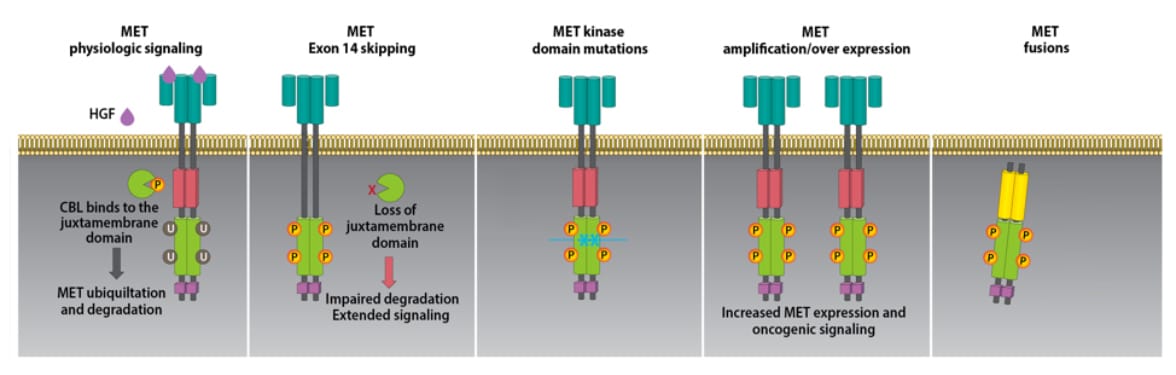
There are 4 major categories of MET protein alterations: Mutation, Amplification, Over Expression and Fusion.
MET Mutation
The most commonly discussed form of mutation is Exon 14 skipping. Exon 14 skipping occurs when the code contained in Exon 14 is skipped over during the creation of the MET protein. Exon 14 contains the instructions for CBL to bind to MET the protein. CBL is required in protein degradation. The lack of CBL binding extends the life of MET and results in excessive tyrosine kinase signaling resulting in extended cell division.
There are other forms of MET mutations where all Exons of the coding are executed in correct sequence but there is an error in the code Tyrosine Kinase. The result of this mutation is excessive tyrosine kinase signaling cell division.
MET Amplification
The human genome typically contains two copies of each gene. The number of genes is referred to as the gene copy number (GCN.) The higher the GCN, the higher number of copies of genes. As the number of MET genes is increased, there is typically increases in the MET protein that can result in higher than normal tyrosine kinase signaling. Higher tyrosine kinase signaling results in excessive cellular division. In summary, the MET gene and protein are normal but too many genes mean too many proteins resulting in excessive tyrosine kinase signaling and excessive cellular division. Gene GCN can also be described as mean MET copy number per cell (mean MET/cell) and the MET copy number per centromere 7 ratio (MET/CEP7). MET amplification is being identified as a mechanism of resistance to EGFR inhibitors such as osimertinib.
MET Over Expression
In Over Expression, there is an excess of protein on the cell surface. Over Expression of MET could be due to mutation or amplification.
MET Fusion
MET Fusion occurs when two genes fuse together resulting in a hybrid gene containing portions of two disparate genes. The resulting protein contains components of two proteins that may stimulate the kinase activity of MET and promote tumor growth through excessive signaling.
Primary versus Acquired Alterations
When lung cancer is first diagnosed, there may be only a single genetic alteration. This is the primary genetic alteration. However, a genetic alteration can also be acquired as a resistance mechanism to treatment. As an example, a person with a primary alteration of EGFR or ALK, who has been on a drug for that alteration, may also later acquire a MET alteration. They will then have two genetic alterations which may be treated in combination.